One bad batch can shut your business down overnight.
In South Africa, food safety laws, medicines control, and ISO standards all demand full product traceability. Regulators don’t accept “We couldn’t find the records.”
Manual spreadsheets break under that pressure. They miss data, create gaps, and fall apart during audits.
Inventory traceability ERP in South Africa solves this. Acumatica tracks every lot and serial number, from the supplier’s batch all the way to your customer’s door.
This guide shows you exactly how it works, and why it is important for your industry.
Summary
Acumatica’s inventory traceability ERP gives South African businesses full lot and serial-number tracking across receipt, picking, packing and dispatch to meet strict regulatory and audit requirements. It automates data capture (batch numbers, expiry dates, certificates, quality holds), enforces FIFO/FEFO picking, and produces instant recall and compliance reports, eliminating spreadsheet gaps and reducing recall time from days to minutes.
Key points
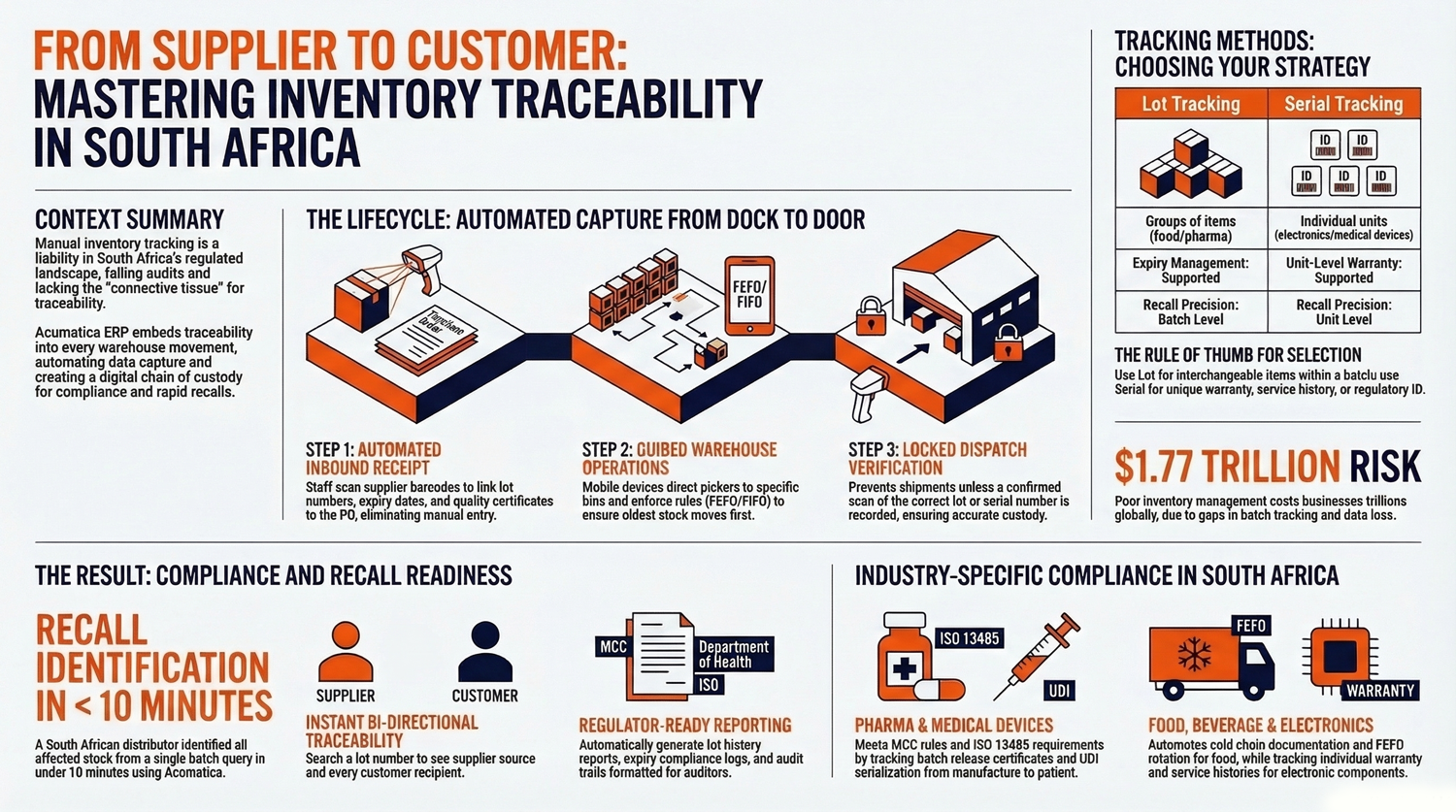
- Lot vs serial tracking: lot tracks groups (food, pharma, chemicals); serial tracks individual units (electronics, medical devices, warranty/service needs). Choose based on interchangeability and regulatory needs.
- Automated capture: scans at receipt and during warehouse operations store supplier batch, expiry, certificates, and quality holds so no manual re-entry or audit gaps.
- Guided warehouse processes: mobile picking, enforced FIFO/FEFO/LIFO, and locked dispatch ensure correct lots/serials shipped and a complete custody record.
- Fast recalls and regulatory reports: query a lot or serial to instantly see upstream supplier info and downstream customer deliveries; generate regulator-ready reports and attachments.
- Industry fit: configurable for South African pharma (MCC), food safety/cold chain, electronics warranty/service, and medical device UDI/ISO 13485 traceability.
Want to see it working? Book a free Acumatica demo with Astraia to review traceability in your industry.
Lot Tracking vs. Serial Tracking, What’s the Real Difference?
Not all products need to be tracked the same way. Some move in groups. Others need their identity. Picking the wrong method means your compliance records won’t hold up when it matters most, and in South Africa’s regulated industries, that’s a serious problem.
Here’s how the two approaches work, and when to use each one.
What Is Lot Tracking?
Imagine you receive 500 boxes of medication from a supplier. They all came from the same production run, same raw materials, same date. That’s a lot, and lot tracking gives that entire group one shared number.
If one box later fails a quality check, you don’t need to guess. You pull up that lot number and instantly see every unit from that batch. Where it came from. Where it went. Who has it now?
This is how batch management works in food, pharma, and chemical distribution across South Africa.
Acumatica captures the following at receipt:
- Supplier batch number
- Expiry date (which triggers automatic FEFO or FIFO picking rules)
- Certificates of analysis linked to the lot
- Quality hold status, so you can freeze one bad batch without touching the rest of your stock
You can also split or combine lots inside the warehouse without losing the traceability trail.
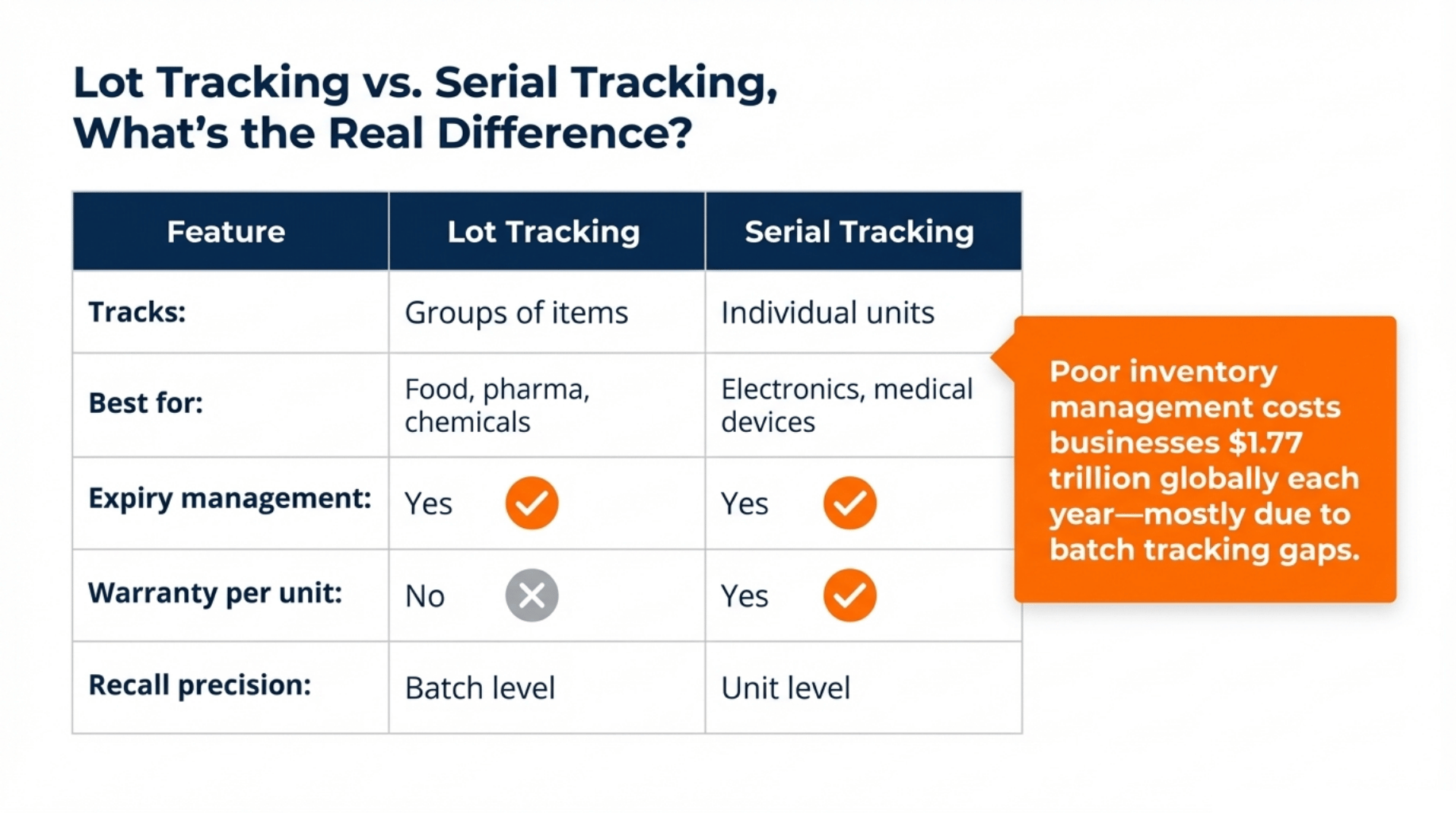
Poor inventory management costs businesses an estimated $1.77 trillion globally each year. Most of those losses trace back to gaps in batch tracking.
What Is Serial Number Tracking?
Serial tracking goes one level deeper. Instead of tracking a group, you track each individual item.
Every unit gets its own unique number at the point of receipt or production. That number stays with the item through picking, dispatch, and delivery, and lives on long after the sale.
Think about a solar inverter or a medical device. The customer needs a warranty. Your service team requires a repair history. Your compliance officer requires proof of exactly which unit went to which customer.
Serial tracking makes all of that possible.
Inside Acumatica, each serial number is linked to:
- The specific sales order and customer
- An automatically calculated warranty expiry date
- A full-service history, repairs, returns, and replacements logged against that unit
No guesswork. No digging through spreadsheets. Just one number that tells the whole story.
Quick comparison:
| Feature | Lot Tracking | Serial Tracking |
|---|---|---|
| Tracks | Groups of items | Individual units |
| Best for | Food, pharma, chemicals | Electronics, medical devices |
| Expiry management | ✅ Yes | ✅ Yes |
| Warranty per unit | ❌ No | ✅ Yes |
| Recall precision | Batch level | Unit level |
The rule of thumb: if items are interchangeable within a batch, use lot tracking. If each unit has its own life, warranty, service, or regulatory ID, use serial tracking.
How Acumatica Captures Traceability Data at Every Step
Traceability only works if the data is captured correctly, every single time. One missed scan, one manually typed number, one skipped step, and your audit trail has a hole in it.
That’s where most businesses fail. They have the intention to track everything, but the process breaks down on the warehouse floor.
Acumatica removes the guesswork. It captures lot and serial data automatically at every touchpoint, receipt, picking, packing, and dispatch. No manual re-entry. No gaps. No excuses when a regulator comes knocking.
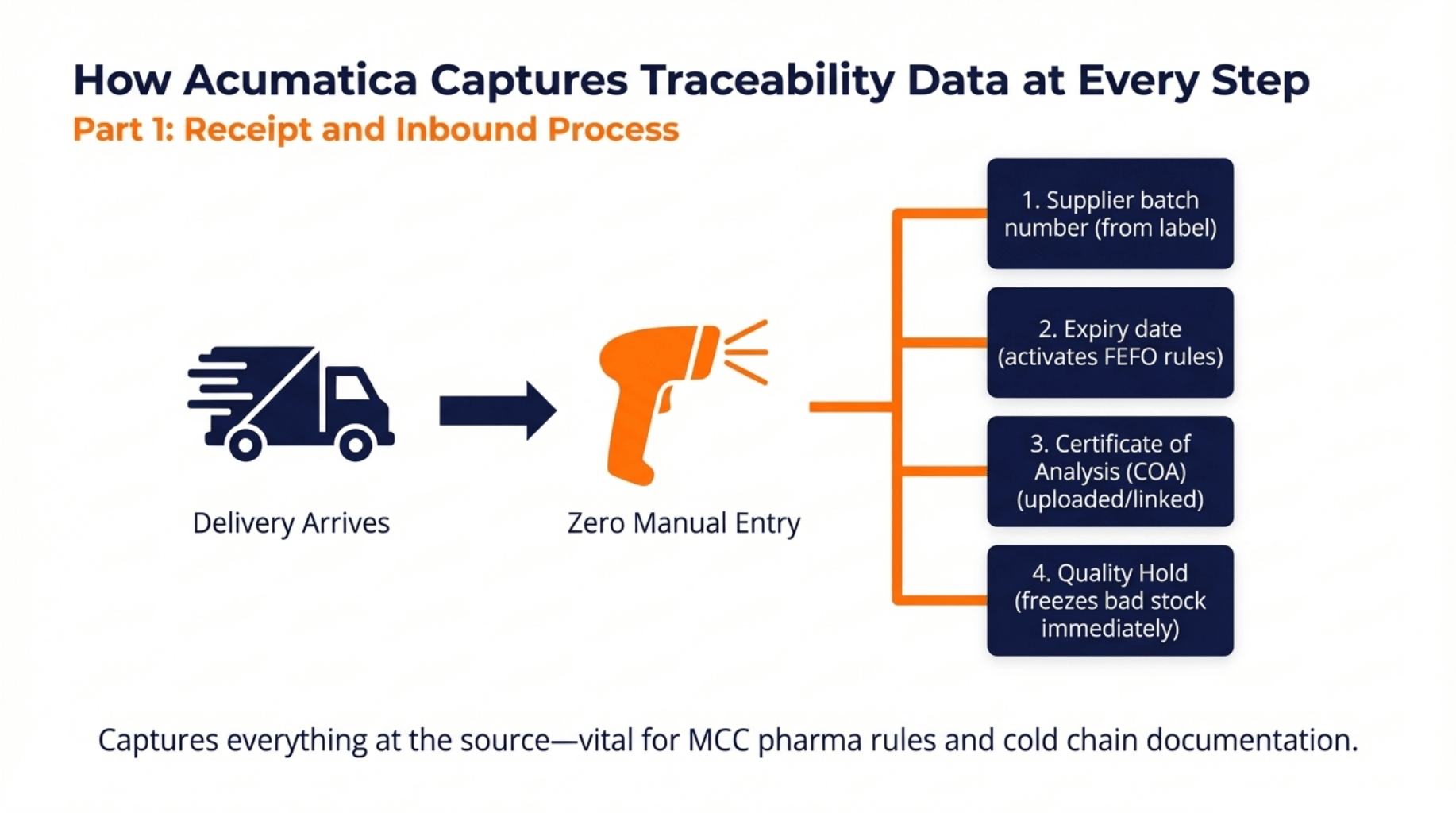
Receipt and Inbound Process
Traceability starts the moment a delivery arrives at your door.
With Acumatica, your receiving team doesn’t type anything manually. They scan the supplier’s barcode and the system does the rest, pulling in the lot number, linking it to the purchase order, and prompting for any additional details like expiry date or certificate of analysis.
Here’s what gets captured at receipt:
- Supplier batch number, scanned directly from the delivery label
- Internal lot number, auto-generated or manually assigned
- Expiry date, once entered, automatically activates FEFO picking rules
- Certificate of analysis, uploaded and linked to that specific lot
- Quality hold, applied before the stock enters available inventory, so nothing ships before it’s cleared
From the first scan, every movement of that lot is recorded. You have a full audit trail before the goods even reach the shelves.
This matters in South Africa’s regulated industries. Whether you’re receiving pharmaceutical stock under MCC rules or food products that need cold chain documentation, Acumatica captures it all at the source.
Picking, Packing, and Dispatch
The warehouse is where traceability usually falls apart.
A picker grabs the nearest box instead of the oldest batch. Two lots get mixed in one shipment. Someone skips the scan because they’re in a rush. By the time you notice, the data is gone.
Acumatica prevents all of this.
When a pick task is created, the system tells the worker exactly which lot to pick, based on your FIFO, FEFO, or LIFO rules. The mobile device shows the bin location, the lot number, and the quantity. The picker scans to confirm. If they try to pick the wrong lot, the system stops them.
What this looks like in practice:
- Guided picking, mobile device directs workers to the exact bin and lot
- Rule enforcement, FIFO, FEFO, or LIFO applied automatically per item class
- Auto-populated documents, packing slips and delivery notes show the lot and serial numbers without anyone typing them
- Locked dispatch, no shipment goes out without a confirmed scan
- Full chain of custody, every hand that touched the stock is recorded
By the time the goods leave your warehouse, Acumatica has a complete record. The customer, the lot, the serial numbers, the date, the picker, all of it is there.
If a problem surfaces later, you don’t spend days reconstructing what happened. You run a report, and you have your answer in seconds.

Product Recalls and Compliance Reporting Made Simple
A product recall without traceability data is a nightmare.
You’re on the phone with a regulator, they want to know which customers received a specific batch, and your team is digging through spreadsheets from three different systems. Every minute that passes increases your liability, and your stress.
This is where inventory traceability ERP in South Africa earns its keep. Acumatica gives you the full picture in seconds, not days.
Instant Recall Identification
Speed is everything when a recall hits.
The moment a issue is flagged, whether it’s a contaminated batch, a failed quality test, or a supplier alert, you need two things fast: where the stock came from, and who has it now.
Acumatica handles both in a single query.
Type in the lot number. The system runs a full trace in both directions:
- Upstream, which supplier delivered that batch, on which date, under which purchase order
- Downstream, every sales order, every customer, every delivery that included stock from that lot
From there, Acumatica auto-generates a recall notification list. You know exactly whom to contact and what to tell them, without anyone manually cross-referencing orders.
A pharmaceutical distributor in South Africa used Acumatica to identify all affected stock from a single batch query in under 10 minutes. What could have become a wide-scale recall was contained quickly because the data was already there, structured, and searchable.
That’s not luck. That’s what proper batch management looks like in practice.
You can also search by serial number or date range, useful when a recall spans multiple lots, or when a customer contacts you about a specific unit they received months ago.
Regulatory Compliance Reports
Regulators don’t want your good intentions. They want documentation.
In South Africa, that means satisfying the Medicines Control Council, the Department of Health, food safety inspectors, and ISO auditors, often all at once. Each body has its own requirements, and the records need to be accurate, complete, and ready on demand.
Manually pulling that information together takes hours. And manual processes introduce errors at exactly the moment you can’t afford them.
Acumatica generates the reports regulators need automatically. Here’s what’s available out of the box:
- Lot history report, every movement from receipt to final delivery, timestamped and linked to the responsible user
- Certificate of analysis attachment, each outbound shipment shows the linked quality document for that lot
- Expiry date compliance report, confirms your FEFO picking rules were followed correctly
- Full audit trail, searchable by lot number, serial number, customer, or supplier
- Electronic records, formatted and accepted by South African regulatory bodies
No scrambling. No reconstructing records from memory. Everything is already captured in the system from the moment stock arrives.
If an ISO auditor walks in tomorrow, you can pull a complete product history in minutes, and hand them a printed or digital report that covers every step of the chain.
That’s the difference between passing an audit with confidence and spending three days in a panic trying to find paperwork that may or may not exist.
Industry-Specific Traceability in South Africa
Every industry has its own rules, and its own consequences for getting it wrong.
Acumatica adapts lot and serial tracking to fit your specific compliance requirements, not the other way around. Here’s how it works across four key sectors in South Africa:
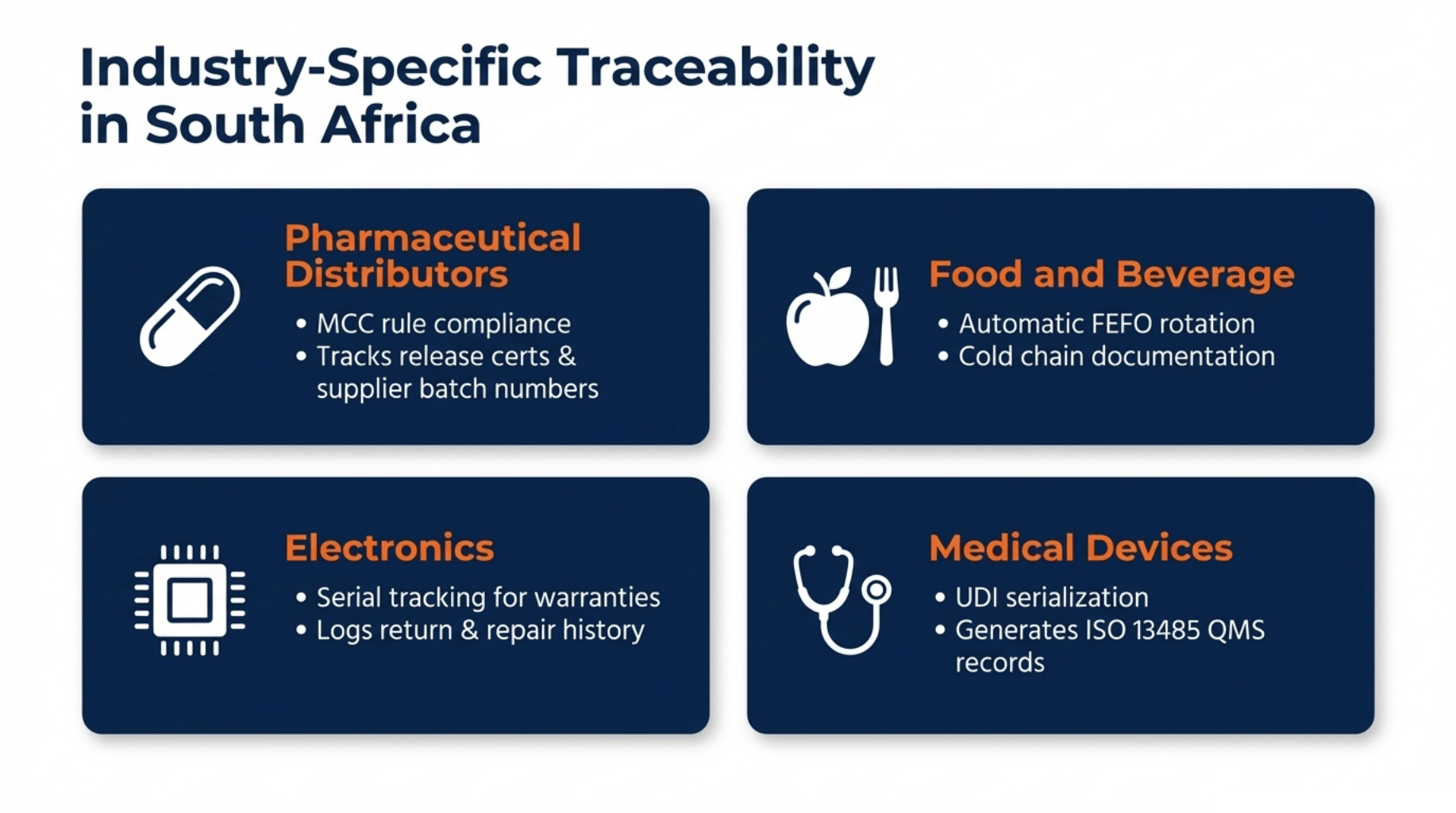
Pharmaceutical distributors
Stock moves under strict MCC rules. Acumatica tracks expiry dates, batch release certificates, and supplier batch numbers for every delivery. If a batch fails, you have the full paper trail ready before the regulator asks.
Food and beverage
Shelf life is everything. Acumatica enforces FEFO rotation automatically, so your oldest stock ships first. Cold chain documentation and food safety audit trails are captured at every step, from the receiving dock to the delivery note.
Electronics
Each unit needs its own identity. Serial number tracking links every device to a customer, a warranty period, and a full-service history. Returns and repairs are logged against the original serial, no guesswork, no disputes.
Medical devices
Patient-level traceability is non-negotiable. Acumatica supports UDI serialisation and generates the ISO 13485 records your quality management system requires. Every device is traceable from manufacture to patient.
Whatever your industry, the principle is the same: one system, full visibility, zero gaps.

Wrapping Up
Full inventory traceability is not a luxury. It’s your protection.
One untracked batch can trigger regulatory action, damage your brand, or land you in a legal dispute you didn’t see coming. South African businesses in pharma, food, electronics, and medical devices can’t afford that risk.
Acumatica gives you the tools to capture, track, and report every lot and serial number, from your supplier’s batch to your customer’s hands.
Ready to see it live? Book a free Acumatica demo with Astraia, and we’ll show you exactly how traceability works in your industry.
FAQ Section
Q1: What is the difference between lot tracking and serial tracking in ERP?
Lot tracking groups multiple units under one batch number. Serial tracking assigns a unique number to every individual item. Both methods are supported in Acumatica depending on your product type.
Q2: Does Acumatica support FIFO, FEFO, and LIFO picking for lot-tracked items?
Yes. Acumatica applies picking rules per item class, automatically directing warehouse staff to the correct lot based on your chosen rotation method.
Q3: How does ERP help with a product recall in South Africa?
Acumatica traces a batch upstream to the supplier and downstream to every customer order in seconds, generating a full recall list and audit report.
Q4: Which industries in South Africa need inventory traceability ERP?
Pharmaceutical, food and beverage, medical devices, electronics, and chemical distribution are the most regulated, but any business needing warranty or quality management benefits.
Q5: Can Acumatica capture lot data using barcode scanners?
Yes. Mobile devices and barcode scanners integrate directly with Acumatica’s warehouse module, capturing lot and serial numbers at receipt, picking, and dispatch.





